Metal Foam Bone Replacements
Why?
Previously, bone replacements were made of solid metal, usually something like titanium. But these solid metal replacements cause stress shielding. Thus, newer bone replacements are made of porous metal, or metal foam. By making the metal porous, the strength of the replacement is decreased, making it have properties more similar to those of bone and therefore reducing the risk of stress shielding.
How?
Metal foam isn't as easy to make as one might think. There are some super technical methods of making it, which probably yield better results, but we don't have to resources to use those methods. Thus, we are making our metal foam using a simple method involving heat and salt.
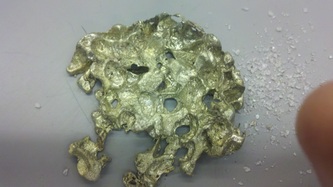
Spring '13: This semester, we used lead-free solder as our metal. First, we heated our solder and salt in separate crucibles in an oven. Then, we dumped our salt into a muffin tin, followed quickly by our molten solder. We stirred it up briefly and then applied a weigh on top of the mixture before letting the whole thing cool. Once everything was cool, we submerged the contents of the muffin tin in warm water to dissolve the salt out of the solder, leaving behind a porous metal structure: our metal foam.
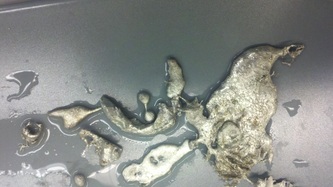
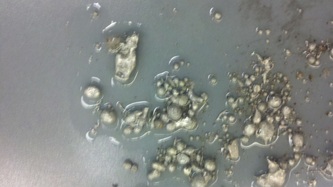
Fall '13: I took several different approaches this semester in an attempt to make metal foams. First, I used zinc as my metal and heated it with an induction coil before pouring into an ingot mold filled with salt. This resulted in just large chunks of zinc with a pocketed bottom surface; the zinc just sat on top of the salt instead of infiltrating it. Next, I tried the same process, but with a extra step to mix the two substances together. This work slightly more successfully, but still not very well. I gave up on this particular method and tried something new: metal pellets. I found some tin pellets and mixing these with the salt directly in the ingot mold, before heating the whole mixture with a pancake coil. This resulted in a porous structure, but because the pellets and salt crystals were large, there was too much empty space, meaning that the actual foam created was very thin once the metal sank into the cracks. My last attempt involved a similar method, but instead of pellets, I used powder. However, I did not have tin powder, only aluminum. Unfortunately, aluminum has a much higher melting point so the pancake coil was not able to fully melt the metal.
Spring '13: This semester, we used lead-free solder as our metal. First, we heated our solder and salt in separate crucibles in an oven. Then, we dumped our salt into a muffin tin, followed quickly by our molten solder. We stirred it up briefly and then applied a weigh on top of the mixture before letting the whole thing cool. Once everything was cool, we submerged the contents of the muffin tin in warm water to dissolve the salt out of the solder, leaving behind a porous metal structure: our metal foam.
Fall '13: I took several different approaches this semester in an attempt to make metal foams. First, I used zinc as my metal and heated it with an induction coil before pouring into an ingot mold filled with salt. This resulted in just large chunks of zinc with a pocketed bottom surface; the zinc just sat on top of the salt instead of infiltrating it. Next, I tried the same process, but with a extra step to mix the two substances together. This work slightly more successfully, but still not very well. I gave up on this particular method and tried something new: metal pellets. I found some tin pellets and mixing these with the salt directly in the ingot mold, before heating the whole mixture with a pancake coil. This resulted in a porous structure, but because the pellets and salt crystals were large, there was too much empty space, meaning that the actual foam created was very thin once the metal sank into the cracks. My last attempt involved a similar method, but instead of pellets, I used powder. However, I did not have tin powder, only aluminum. Unfortunately, aluminum has a much higher melting point so the pancake coil was not able to fully melt the metal.